Creating the blueprint
for medicines on demand.
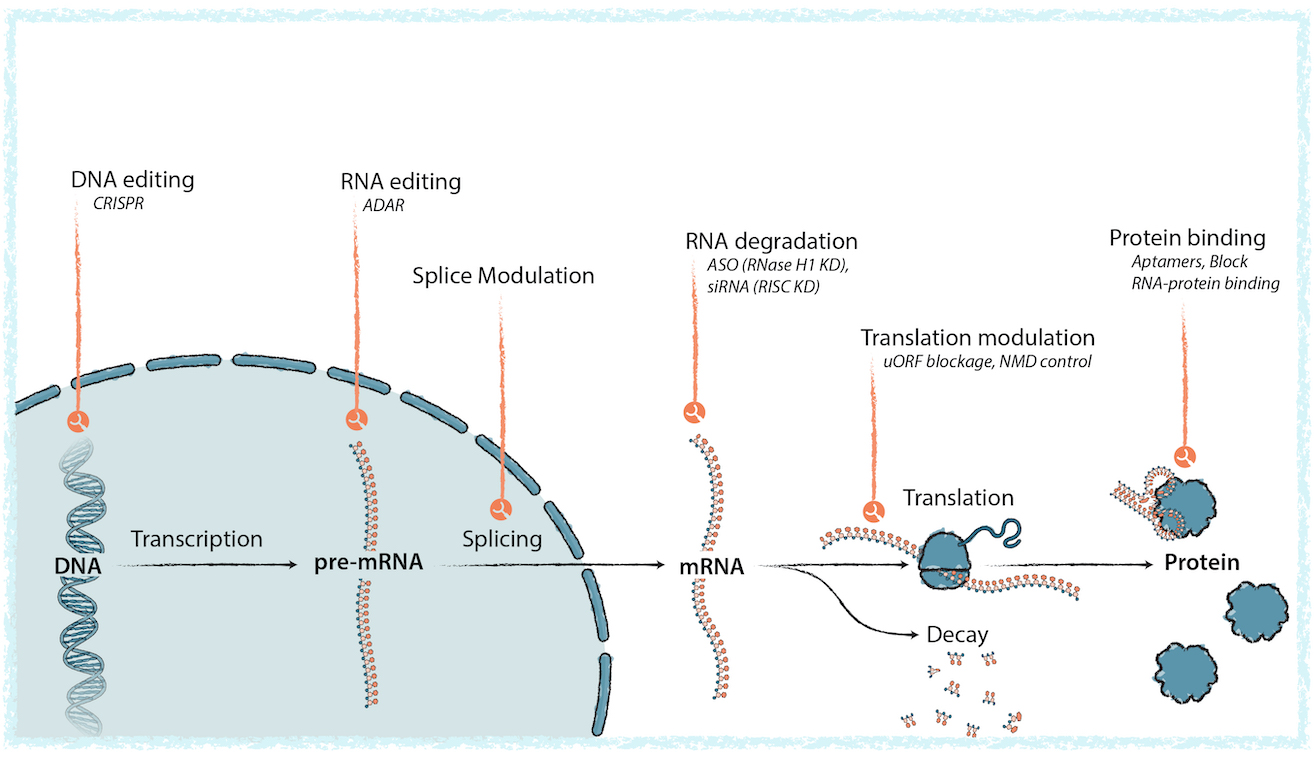
OBMs are an ideal modality for creating gene-centric therapies. Unlike other drug modalities such as small molecules or antibodies, a principal way OBMs interact with targets is through well understood Watson-Crick-Franklin hybridization: a zipper-like process that selectively and precisely targets specific positions of DNA or RNA molecules within the cell. They can control the effective activity of genes through multiple mechanisms. They can enhance or block molecular interactions which can control how genetic information is used by the cell. This provides the foundation for creating new medicines which directly address the genetic and molecular basis of disease – from preventing the production of toxic RNAs or protein products, to increasing the levels of insufficiently expressed enzymes, to even editing mRNA or changing how the splicing machinery assembles the genetic instructions which control protein production. Creyon’s technologies and efforts make engineering new OBMs to treat disease through these varied mechanisms, and perhaps yet to be discovered mechanisms, not only possible, but fast and efficient. We are even engineering delivery systems to directly target OBMs to specific cell types and tissues.
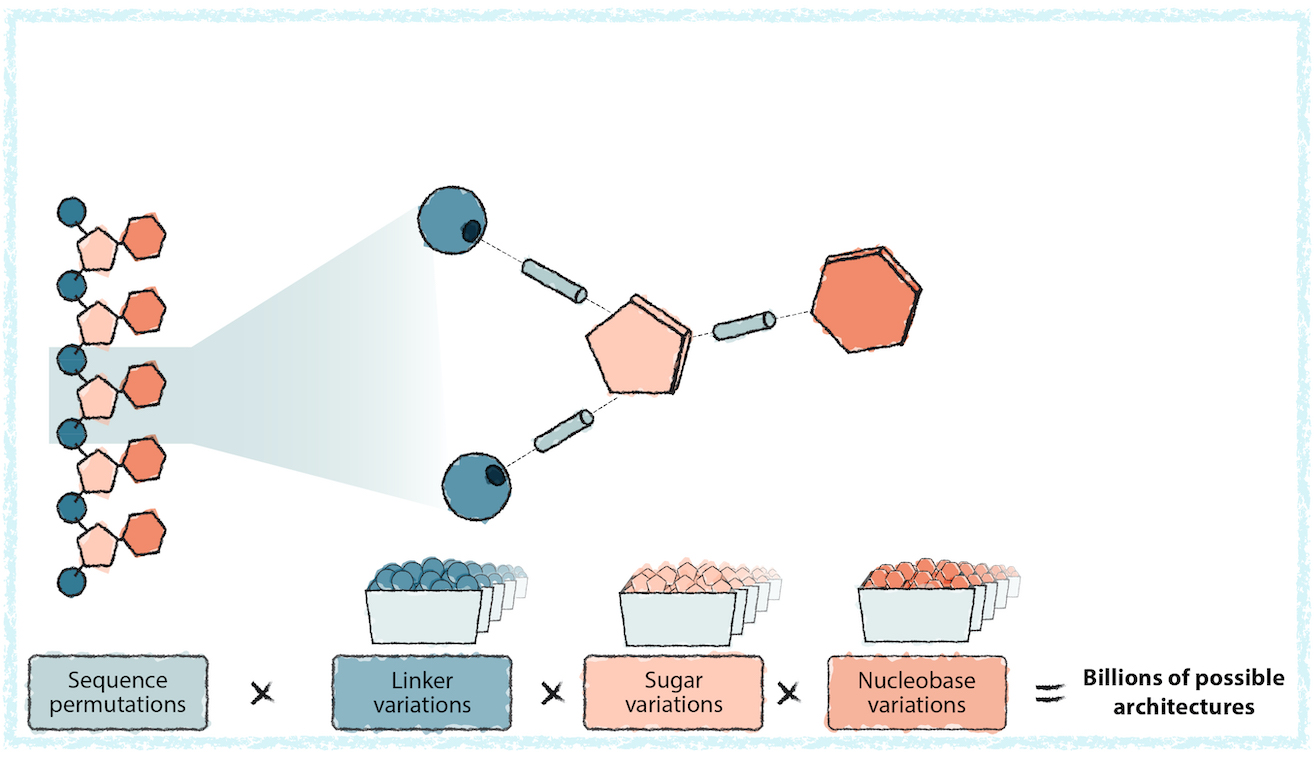
But the sequence of an OBM is just one aspect of what makes it work. From the sequence composition itself to the sugar, nucleobase, and backbone chemistries, every component is modifiable and can have critical implications for its pharmacology, from its safety and efficacy to how long its effects will last. Yet there are 4 billion possible sequence configurations in a molecule that is just 16 bases long. Add a few sugar and backbone modifications, and that number skyrockets to more than billions of possible designs — an impossibly large number of possibilities that could never be screened or explored with traditional methods. The Creyon™ Platform creates the right data to explore this space far more efficiently and develop predictive models that integrate learnings across hundreds of thousands of data points to create new medicines that are knowably safe and effective.
Creyon’s groundbreaking approach is upending the process of drug development by uncovering the engineering principles that allow us to employ the right designs right away. With a deliberate approach to engineering medicines instead of trial-and-error screens, Creyon is engineering molecules that can be precisely tuned with predictable results before they’re even tested.
